The 3 Stages of Type 1 Diabetes: Why Early Screening and Detection Matter
Hali Broncucia, Research Scientist at the Barbara Davis Center for Diabetes • March 16, 2026
TL;DR
Type 1 diabetes develops in three stages, beginning long before symptoms appear. Research led by TrialNet and scientists at the Barbara Davis Center for Diabetes shows that screening for diabetes-related autoantibodies can identify the disease years before diagnosis. Early detection can help reduce the risk of dangerous complications like Diabetic Ketoacidosis and allows patients to participate in prevention trials. In 2022, the FDA approved Tzield, the first therapy shown to delay the onset of clinical Type 1 diabetes, highlighting the importance of early screening for people with a family history of the disease.
Why Type 1 Diabetes Often Appears Suddenly
For most people, the onset of type 1 diabetes (T1D) seems to occur suddenly, often resulting in a trip to the emergency room with life-threatening complications such as diabetic ketoacidosis (DKA). The Barbara Davis Center at The University of Colorado, Anschutz is a Clinical Site of TrialNet, a worldwide leader in T1D prevention research and one of the prominent organizations working to change that scenario.
Understanding the 3 Stages of Type 1 Diabetes
In 2015, Breakthrough T1D (formerly JDRF), the
American Diabetes Association (ADA), and the Endocrine Society recommended adoption of a new T1D staging classification (Diabetes Care 2015). This recommendation is largely based on two decades of
TrialNet research involving more than 150,000 relatives of people with T1D. T1D is now most accurately understood as a disease that progresses in three distinct stages (Figure 1).
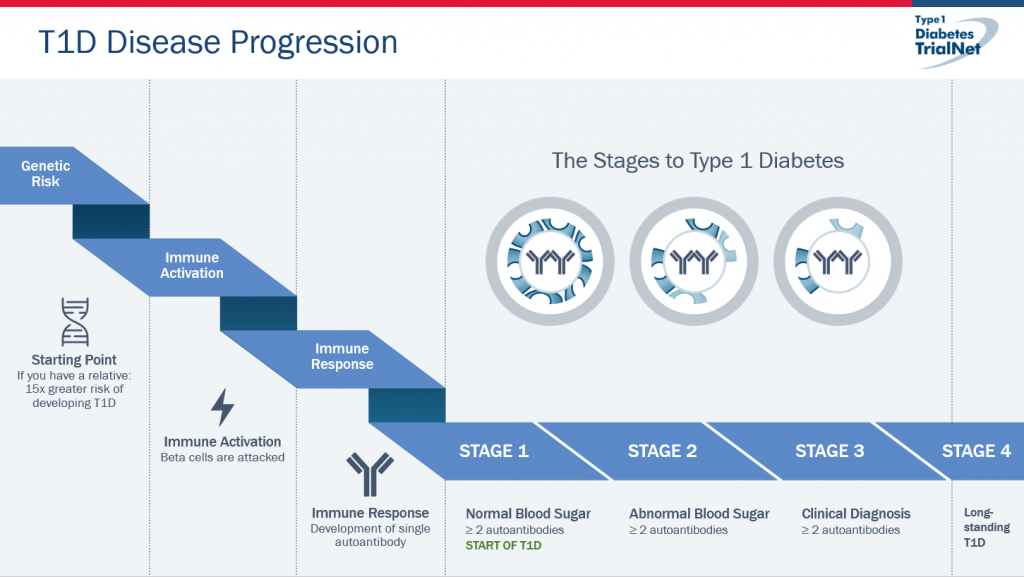
Breaking Down the Three Stages of Type 1 Diabetes
- Stage 1 is the start of T1D. Individuals test positive for two or more diabetes-related autoantibodies but have no symptoms and blood sugar remains normal.
- Stage 2 includes autoantibody positive individuals who have now abnormal blood sugar levels with no diabetes symptoms. For early-stage T1D (stages 1 and 2), lifetime risk of developing clinical T1D approaches 100 percent.
- Stage 3 is the clinical diagnosis of T1D. Individuals generally show symptoms of T1D, including frequent urination, excessive thirst and weight loss.

Why Early Detection of Type 1 Diabetes Is Important
Clinical research supports the usefulness of diagnosing T1D early—before beta cell loss advances to stage 3. The earlier a diagnosis is made in the disease process (early-stage T1D), the sooner prevention therapies can take place, and the more beta cells are likely to remain. More beta cell preservation has been associated with better outcomes regarding blood sugar control and reduction of long-term complications. In November 2022,
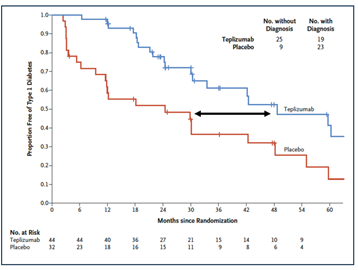
teplizumab (Tzield) was approved by the FDA as the first drug delaying onset of Stage 3 T1D in adults and pediatric patients aged 8 years and older with Stage 2 T1D. It was a TrialNet prevention trial (TN10) that led to this first FDA approved drug to delay the onset of Stage 3 T1D. The study was published in 2019 and showed a 2-year delay of clinical (stage 3) T1D in those at Stage 2 T1D (New England Journal of Medicine, 2019, Figure 2).
How TrialNet Screening Helps Identify Risk for Type 1 Diabetes
Led by Dr. Andrea Steck, the TrialNet team at the Barbara Davis Center is dedicated to preventing T1D and stopping disease progression by preserving beta cells before and after new onset diagnosis. Our Center has multiple ongoing clinical trials in both early-stage T1D (prevention trials) and new onset diabetes (intervention trials).
For people who participate in T1D screening and monitoring research like TrialNet, the risk of DKA at diagnosis decreases from 50 percent to less than 5 percent in our Colorado population (< 18 years at diagnosis).
Who Should Be Screened for Type 1 Diabetes
Both the ADA and Breakthrough T1D recommend screening for people who have relatives with T1D. Family members have a 15 times greater risk of being diagnosed than a person with no family history. Screening is available at no charge for:
- Anyone between the ages of 2 and 45 with a sibling, child or parent with T1D
- Anyone between the ages of 2 and 20 with a sibling, child, parent, cousin, uncle, aunt,
niece, nephew, grandparent, or half-sibling with T1D
- Anyone between the ages of 2 and 45 years who has tested positive for at least one
T1D related autoantibody outside of TrialNet
It is recommended that children who do not test positive for diabetes-related autoantibodies continue to get rescreened every other year until age 18.
Type 1 Diabetes Research at the Barbara Davis Center
The Barbara Davis Center at The University of Colorado, Anschutz offers TrialNet screening and intervention studies. For further information, contact the Prevention Team at 720-346-1034 or hali.broncucia@cuanschutz.edu for any questions or if you would like additional information. Learn more at DiabetesTrialNet.org.
About the Author
Hali Broncucia is a type 1 diabetes Research Scientist in Pediatrics at the Barbara Davis Center for Diabetes on the CU Anschutz Medical Campus. Broncucia has contributed to
several medical publications and currently leads a clinical research team conducting sponsored type 1 diabetes clinical trials.